
Alkaline metal ions like calcium (Ca2+) and magnesium (Mg2+) play a critical role during the process of bone healing. For this reason, Mg2+ and Ca2+ ions are gaining interest in tissue engineering as compounds to be delivered directly at the injury site to support bone regeneration. To meet this need, a team of researchers from The Chinese University of Hong Kong have proposed a novel hydrogel nanocomposite, which incorporates and releases Mg2+ ions with a precise degradation rate, along with a designed intake of Ca2+ ions, for local bone formation and calcification (Acta Biomaterialia 64 389).
A hydrogel design challenge
Researchers have tested different biomaterials to deliver ions at specific injury sites. Among them, hydrogels seem the most appealing for bone healing therapies. This is due to their ability to constitute bone-mimicking, three-dimensional structures that can be loaded with other fillers and injected or shaped in specific geometries.
Magnesium and calcium ions, for example, may be loaded in hydrogels, due to their active role in calcification and bone healing. Calcium intake and deposition, in fact, are fundamental mechanisms during growth and skeletal development, while Mg2+ ions have been shown to enhance cell adhesion and differentiation, along with local bone formation.
However, depending on the interaction between hydrogels and their fillers, some mixtures may be poorly stable, leading to quick degradation and uncontrolled release of fillers. This instability can be detrimental, especially if the retention and release of nanoparticles needs to meet specific requirements for clinical applications. High levels of magnesium in blood, for example, can induce muscular paralysis and hypotension, while an excessive local concentration of Mg2+ ions can cause local bone loss.
A novel design
A Chinese team of researchers, led by Liming Bian, has solved this issue of instability by proposing a nanocomposite in which Mg2+ ions are linked to bisphosphonate nanoparticles (Ac-BP-Mg NPs), which in turn behave as cross-linkers between the polymer chains of a hyaluronic acid-based hydrogel (MeHA, see the zoomed region in the figure).
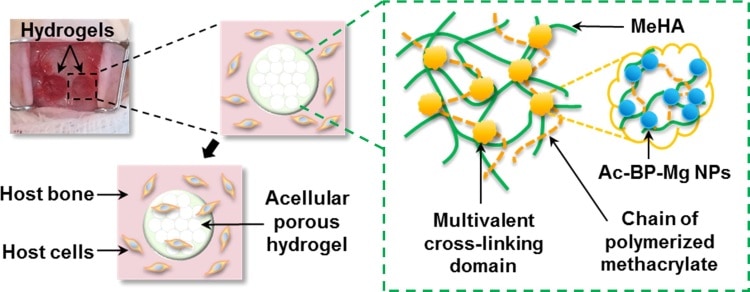
With the aid of this design, the chemical bonds between Mg-containing nanoparticles and the hydrogel network allow nanoparticles to be physically and chemically immobilized, extending the storage time of Mg2+ ions within the network and slowing down their release over time.
Moreover, since bisphosphonates are known to prefer binding to calcium more than magnesium ions, when these hydrogels are implanted in calcium-rich environments – like a bone defect – Mg2+ ions are forced to leave the nanoparticles to be substituted by Ca2+. This creates a magnesium release/calcium deposition cascade, which stimulates bone healing and calcification, simultaneously.
Design in action
The researchers tested the novel hydrogels as two- and three-dimensional substrates for culture of human mesenchymal stem cells. They observed enhanced cell adhesion and spreading on Mg-containing hydrogels, along with osteogenic differentiation when compared with Mg-free controls.
Interestingly, they also tested Mg-containing nanocomposites as acellular implants – i.e. without containing any cells – in rat models presenting skull bone defects. The authors reported colonization by bone cells from the surrounding tissue and new bone formation eight weeks after the implantation. Considering the results, this nanocomposite offers a reliable and appealing proof-of-concept study to engineer future ion-containing hydrogels for tissue engineering applications.



