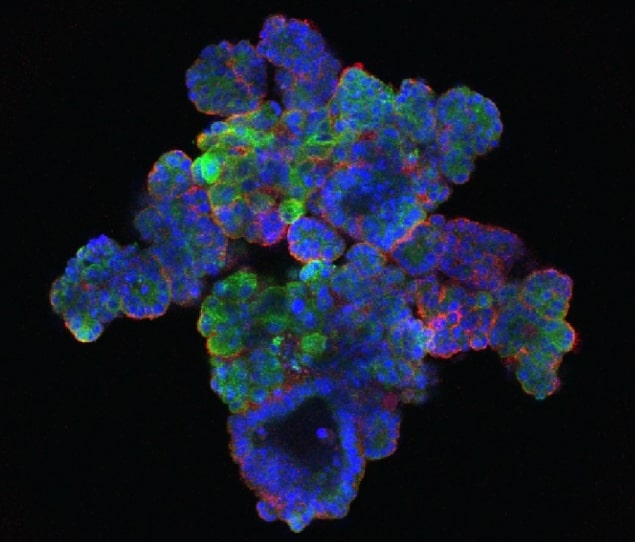
Researchers at the University of Cambridge have developed human liver cancer organoids – small three-dimensional (3D) versions of the human cancer grown in the laboratory – which behave the same outside of the human body as they would inside. The researchers isolated liver cancer organoids from patient samples to successfully grow seven “tumouroid” lines for about one year in culture without losing the cancer’s characteristics (Nature Medicine 23 1424).
Even after months of being cultured outside of the human body, the human tumouroids exhibited their original metastatic potential when transplanted into a mouse. This 3D culture method is an attractive alternative to using animal models, which are expensive and highly time-consuming. These 3D models are also preferable to currently used two-dimensional models, in which cells start behaving very differently from how they would inside the body and change genetic profiles drastically from the original cancer.
The tumouroids expressed genes that corresponded well with those in the cancer from which the line originated. Cancers are known to develop random genetic alterations, which can differ from patient to patient – these tumouroids preserved each individual patient’s cancer-related genetic profile.
Correctly assessing genetic alterations is important in patient-specific therapies and in establishing prognosis of the disease. The researchers demonstrated the effective use of the model by identifying several genes associated with liver cancer that had not been reported before. Furthermore, they found that the overexpression of four particular genes (C19ORF48, UBE2S, DTYMK and C1QBP) was associated with poor-survival prognosis.
The team also used the tumouroid lines as a drug-screening platform against 29 anti-cancer drugs that are currently in clinical use or under development. They applied the platform to identify patient-specific drug sensitivities. While many tumouroids were found to be resistant to the majority of the drugs, some were found to be sensitive to drugs based on their specific genetic alterations.
The researchers have developed a valuable 3D model – by retaining the characteristics of the original cancer, the tumouroids can effectively be used for liver cancer research, drug screening and discovery of new biomarkers for drug development. This novel system could enable personalized medicine in liver cancer treatment.