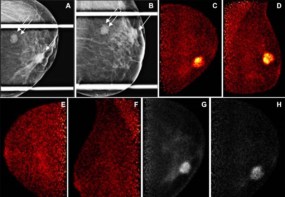
Most solid tumours are poorly oxygenated, rendering them more resistant to both chemo- and radiotherapy. Systemic delivery of oxygen prior to treatment has proven largely ineffective in reversing this resistance. Now, US researchers have shown that bursting oxygen-filled microbubbles injected into breast tumours makes the tumours three-times more sensitive to radiotherapy, improving survival in animal models of the disease (Int. J. Radiat. Oncol. Biol. Phys. doi: 10.1016/j.ijrobp.2018.01.042).
“Finding a way to reverse oxygen deficiency in tumours has been a goal in radiation therapy for over 50 years,” said senior author John Eisenbrey, from Thomas Jefferson University and the Sidney Kimmel Cancer Center. “We’ve demonstrated here that oxygen microbubbles flush tumours with the gas, and make radiation therapy significantly more effective in animal models.”
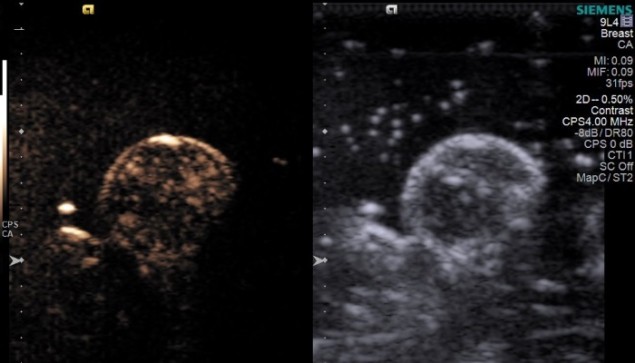
In the study, Eisenbrey and colleagues delivered oxygen-filled microbubbles into the animal’s blood flow via intravenous injection. The bubbles were burst locally, however, only raising the oxygen level within the tumour itself. They demonstrated that the microbubbles successfully and consistently increased breast tumour oxygenation levels by 20 mmHg, significantly more than control injections of saline or untriggered oxygen microbubbles.
The researchers showed that bursting the microbubbles with ultrasound immediately prior to irradiation almost tripled the radiosensitivity of the tumour. It also nearly doubled survival times in the mice – from 46 days with placebo, nitrogen-filled microbubbles, to 76 days with oxygen-filled microbubbles. Using photoacoustic imaging, the investigators also observed that oxygen increased throughout the cancer mass, even in areas without direct access to blood vessels.
“The very act of bursting these microbubbles within the tumour tissue seems to change the local physiology of the tumour and make cells generally more permeable to oxygen and potentially to chemotherapy as well,” explained Eisenbrey. “We think this is a promising approach to test in patients to amplify the effects of radiation therapy.”