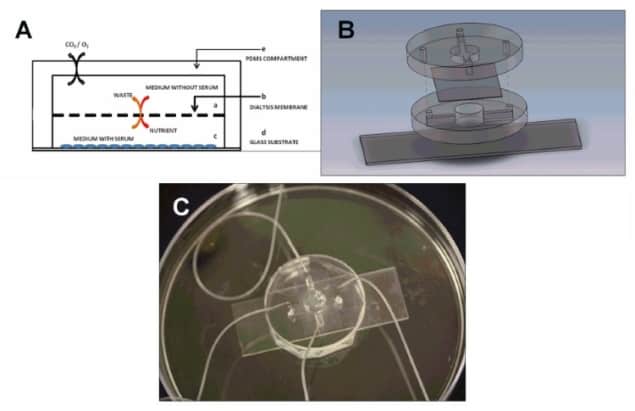
A three-dimensional (3D) bone-on-a-chip with the potential to study cancer metastasis has been developed by researchers at The Pennsylvania State University. The bone-on-a-chip was designed within a microfluidic device – a miniaturized chamber that is continuously fed with nutrient-containing fluids to support the growth of micron-scale bone tissue (Small doi: 10.1002/smll.201702787).
The device demonstrated the ability to support the attachment, growth and spontaneous differentiation of osteoblasts (bone-forming cells) for up to 30 days. The researchers grew the cells on glass inside a cell culture chamber, which received nutrient-containing medium through a dialysis membrane. They observed that the forming bone tissues were able to mature and produce sufficient amounts of alkaline phosphatase (ALP) enzyme and collagen proteins, and accumulate hydroxyapatite minerals – all important for forming bone with structural integrity and functional relevance to real bone.
After maturation of the bone tissue, the team introduced breast cancer cells, mimicking the metastasis of breast cancer to bone. The researchers found that some breast cancer cells were dormant (or quiet) in the 3D bone environment, without any invasive behaviour and showing no noticeable effect on the bone.
On the other hand, they saw that aggressive breast cancer cells were highly invasive in the 3D bone. These cancer cells dug themselves into the bone by eroding the collagen and consuming the bone matrix, resulting in the formation of large holes in the 3D bone. After two weeks, the aggressive breast cancer cells had integrated themselves into the bone.
The 5-year survival rate of patients with breast cancer drops below 30% when the cancer spreads to other parts of the body, with 70% of such metastasis travelling to bone. Once cancer finds its way to bone, it is virtually incurable. For this reason, it is imperative to understand the interactions between cancer cells and bone. The researchers in this study constructed a model that can help reveal the mechanisms behind bone metastasis. With better understanding of the disease using such platforms, therapeutic targets could be developed to treat and prevent metastasis from occurring.